|
1/7/2024 0 Comments Acetate molar massA practical treatise on the manufacture of vinegar. ^ Australia New Zealand Food Standards Code "Standard 1.2.4 - Labelling of ingredients"."Preparation of single cells for imaging/profiling mass spectrometry". You must also remember the relative atomic mass (Ar) of each constituent element. Ullmann's Encyclopedia of Industrial Chemistry. Ethyl acetate can be represented by the molecular formula CH3COOCH2CH3. Journal of the American Chemical Society. ^ a b c d "Safety Data Sheet of Ammonium Acetate" (PDF).Obtaining crystalline ammonium acetate is difficult on account of its hygroscopic nature. Production Īmmonium acetate is produced by the neutralization of acetic acid with ammonium carbonate or by saturating glacial acetic acid with ammonia. It is approved for usage in Australia and New Zealand. a Game of Molecular Construction Simplifying the complex chemical reactions. Molecular weight of Barium Acetate Barium Acetate molecular weight Molar mass of Ba (C2H3O2)2 255.41504 g/mol Convert grams Barium Acetate to moles or moles Barium Acetate to grams Molecular weight calculation: 137.327 + (12.01072 + 1.007943 + 15. part of calley's method for lead artifact conservationĪmmonium acetate is also used as a food additive as an acidity regulator INS number 264. In this lab, the mass of vinegar was 100 g before adding it to container A.a reagent in agricultural chemistry for determination of soil CEC ( cation exchange capacity ) and determination of available potassium in soil wherein the ammonium ion acts as a replacement cation for potassium.a protein precipitating reagent in dialysis to remove contaminants via diffusion. It can be calculated by adding the invididual molar mass of every atom that are composing the molecule (CH4 : 1 atom of C -> molar mass 12.011 g/mol + 4 atom of H -> molar mass 1.008 g/mol -> molar mass of CH4 12.011+41.00816.a catalyst in the Knoevenagel condensation and as a source of ammonia in the Borch reaction in organic synthesis.density of potassium acetate is equal to 1 570 kg/m at 25C (77F or 298.15K) at standard atmospheric pressure. 
In SI, the unit for molar mass is kg/mol. In other words, it is the mass of one mole of a particular substance. The molar mass is a physical property, which is defined as the mass of a substance divided by its amount of substance in moles. Potassium acetate weighs 1.57 gram per cubic centimeter or 1 570 kilogram per cubic meter, i.e. One mole of pure carbon-12 has a mass of exactly 12 grams. Other volatile salts that have been used for this include ammonium formate. Equivalent molar concentration per liter. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set). It is also popular as a buffer for mobile phases for HPLC with ELSD detection for this reason. The molar mass of C3H6O2 (Methyl acetate) is: 74.079 grams/mol. 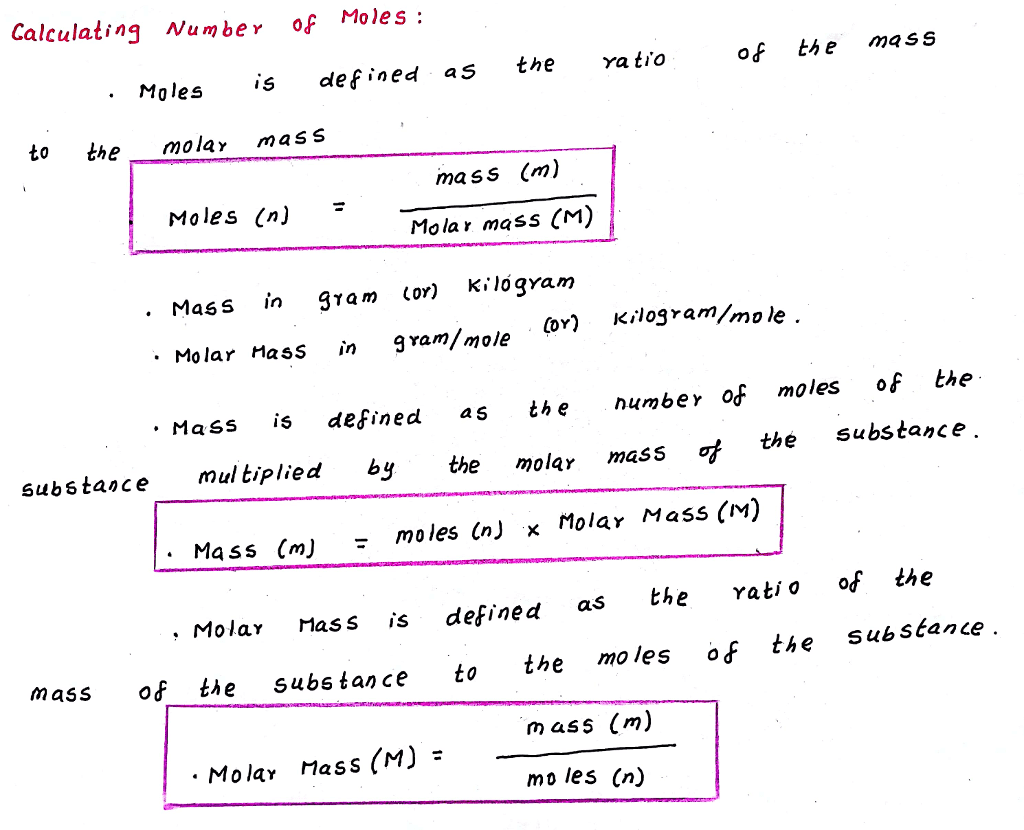
Because of this, it has been used to replace cell buffers that contain non-volatile salts in preparing samples for mass spectrometry. Ammonium acetate is volatile at low pressures. Buffer Īs the salt of a weak acid and a weak base, ammonium acetate is often used with acetic acid to create a buffer solution. It is the main precursor to acetamide: NH 4CH 3CO 2 → CH 3C(O)NH 2 + H 2O
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
